Acid dyes, the salt of sulphuric or carboxylic acid are normally applied from acidic medium and so get the name “acid dye”. Acid dye for nylon is a good choice. By controlling pH, heat, retarding agent, uniform dyeing can be achieved by acid dye for nylon.
Though acid dyes having poor wash fastness, the light fastness property is pretty good. Acid dye has affinity for protein fibers and wool, silk and nylon are often dyed with it.
Dyeing mechanism of acid dye
Polyamide fibers are comparatively stronger than other common fibers. The molecular chains in polyamide are linear and unlike wool, have no side chains with dye sites. The dye sites in polyamides for acid dyes are amino groups, which terminate the molecular chains.
The sulphonic acid group of acid dye can interact with one amino group present in the fiber. The dyeing mechanism of nylon polymide is identical with wool dyeing with acid dyes.
Here, ionic bond is formed between protonised amino group of fiber and acid group of dye to retain dye with fiber.
Nylon has –NH2 group in its’ structure. In acidic bath, the amino group of the fibers are protonated and produce –NH3+. The absorption of dyes by the fiber increases with time, until it reaches to a specific value called equilibrium sorption and thereafter remains unchanged.
Ctionic nature of the nylon fiber increases with the acid treatment. And it produces more and more positive sites (-NH3+) in the fibers through sorption of excess acid.
When an acid dye (R-SO3Na) is added to the bath that contains cationic nylon, the dye anion gets attached with cation of the fiber through electrostatic force with liberation of salt. Some of the group of the fiber bound to the dye molecule with hydrogen bond also.
Generally, the affinity of an acid dye for nylon is greater than wool. Thus, during dyeing of nylon with acid dyes need special care to achieve uniform dyeing.
Uniform dyeing can be possible by pH control, slow uniform heating of the dye bath, and perfect use of retarding or levelling agent in the dye bath. Strong concentrated acid may damage or weaken the fiber.
Polyamide is subjected to chemical damage at low pH so dyeing below pH 3 is not convenient. In case of drying, excessively high heat about 170℃ may impart yellowing effect on the fibers. After dyeing fibers need after-treatment to improve color fastness.
To know acid dye mechanism for wool briefly click here
Acid Dye For Nylon
Nylon can be easily dyed with acid dye. So, how can you dye your nylon substrate with acid dye? Here are the steps:
Typical Recipe
Acid dyes – X%
Wetting agent – 0.5 – 1 g/L
Levelling agent – 1 – 2 g/L
Anti-creasing agent – 1 – 2 g/L
Ammonium sulphate – 2 – 4 g/L (For light shade, pH range 6 – 7)
Or, Sodium acetate – 1 – 2 g/L
Acetic acid – to control pH (For medium to deep shade, pH range 6 – 7)
Temperature – 100℃
Time – 30 – 50 min
M : L – 1:10
Functions of the chemicals
- Wetting agent – By reducing surface tension of water, it helps to get wet the fabric. It enables the water droplets to spread onto a film. By lowering the surface tension, it lowers the energy required to spread the droplet onto a film.
- Acid dye – The main coloring component
- Acetic acid – To control the pH at acidic medium ( 4.5 – 6 depending on shade)
- Levelling agent – Used for level dyeing as super milling dye posses poor levelling property due to higher affinity for fiber
- Sodium acetate & acetic acid – used as Buffer
Procedure
- Set dye bath with substrate at 40℃
- Add wetting agent, leveling agent and buffer (sodium acetate & acetic acid for maintaining pH 4.5 – 6)
- Run the bath for 10 min at same temperature
- Add dyes and run for 10 min again
- Raise the temperature to 95-100℃ over 30-45 min
- Run the bath at 95-100℃ for 30-50 min
- Cool down the bath to 60℃ @ 2-3 /min
- Drop the bath & go for the after-treatment process
After-treatment
- Rinse with hot water and then do follow by cold water rinsing process.
- For improvement of polyamide or Nylon dyeing with acid dyes can improve by treating with a suitable fixing agent at 60 – 70 ℃ for 15 – 20 min at 4 – 5 pH level (With acetic acid)
- Rinse again to clean unexpected chemicals any more
Dyeing Curve
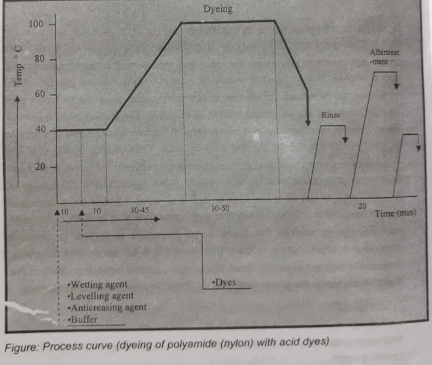
Image Source: Practice of Textile Coloration
Wrapping It Up!
Acid dye for nylon, is not only a solution for nylon. All of the protein materials can be dyed with acid dyes.
You can also check: Acid dye [A brief look]
Salma Hasin the author of this site completed her BSc. in Textile Engineering (Wet Processing Engineering). She wants to share her knowledge to help students in their studies and businessman & entrepreneurs in their businesses in making wise decisions fast.

